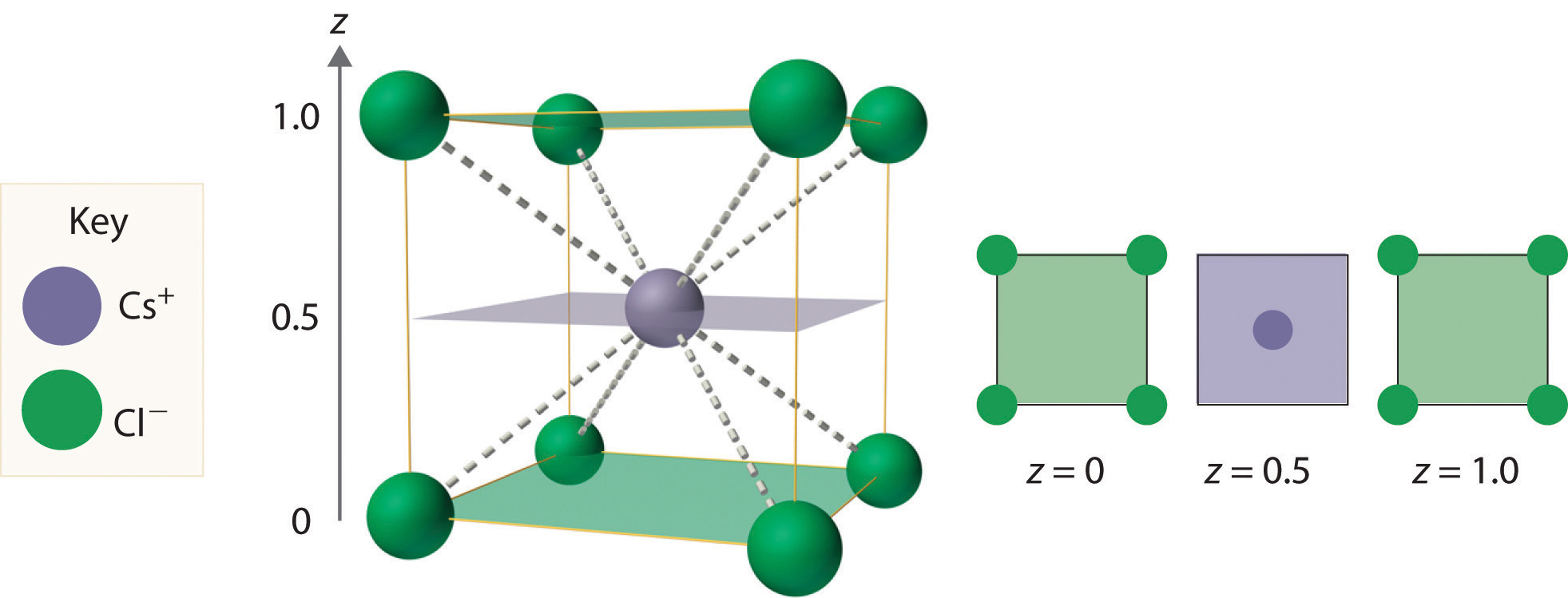
Let us find out the F.C of CaF 2 lewis structure.ĬaF 2 lewis structure has an overall Zero formal charge. The formal charge is an indicator of this stability. Lewis structure stability is based on the internal charges of an atom or molecule. The final lewis structure of the CaF 2 molecule is shown in the image below: Lewis structure of the CaF 2 CaF 2 lewis structure formal charge One electron is transferred from the central Ca atom to the two iodine atoms.It displays an incomplete octet since it is surrounded by just 4 bonding electrons.The calcium atom, which is the core atom in the CaF 2 Lewis structure, is joined to the fluorine atoms by two single bonds Ca-F.Determine the stability of octet by valence electron in CaF 2 : As CaF 2 is not a covalent compound, its particular structure can not be drawn. Fluorine on the other hand surrounds the calcium and has a negative charge. The central atom in the CaF 2 lewis structure is Calcium with a 2+ charge. The complete electron pair is determined by dividing the valence electrons on CaF 2 by 2, which is 16 / 2 = 8. By using the periodic table, it will be noted that the total valence electron in CaF 2 is 16. 
In this step, calculate all the valence electrons present in the CaF 2 molecule. How to draw CaF 2 lewis structure?Ī Lewis structure is a schematic representation of the valence electrons in a molecule represented by lines and dots. Below is further detail about the CaF 2 lewis structure, hybridization, the octet rule, valence electron, and several other facts. CaF 2 has a density of around 3.1g/cm 3.ĬaF 2 is solid, it is colourless and insoluble. The molar mass of the Lewis structure of CaF 2 is 78.07 g/mol. CaF 2 exists as a single transparent crystal that is white in appearance. Let us take a look at some facts about CaF 2 lewis structure in detail.Ĭalcium fluoride, also known as CaF 2, is an inorganic substance comprised of calcium and fluorine atoms. CaF 2 is a chemical compound with different properties in chemistry.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
